Cell | Research Group led by Prof. LI Dapeng Deciphers Complete Biosynthesis of Nicotine
Nicotine, a potent insecticidal alkaloid unique to the nightshade family, has been employed in agriculture as a pesticide since 1690. It also holds therapeutic potential for neurological disorders such as Alzheimer’s, Parkinson’s, and depression. Yet, despite its profound influence on human history, agriculture, and plant evolution, the final steps of nicotine biosynthesis remained an enduring scientific enigma for nearly 80 years following its initial isolation in 1828.
On April 1st, 2026, a research group led by Prof. LI Dapeng from the Center for Excellence in Molecular Plant Sciences, Chinese Academy of Sciences, published a landmark study in Cell titled “Complete Biosynthesis of Nicotine.” Using an innovative information theory-guided multidimensional omics approach, the team fully elucidated the complete biosynthetic pathway of nicotine in Nicotiana attenuata—the very wild tobacco species used by Indigenous peoples thousands of years ago. Their findings reveal a five-component dynamic metabolon that orchestrates the final condensation steps of nicotine synthesis and its transport, solving a long-standing puzzle in plant specialized metabolism and plant defense.
The research began with the unexpected discovery of a nicotine-free mutant, ao2, in wild tobacco, which led to the identification of the key gene NaAO2, essential for the formation of nicotine’s pyridine ring. By constructing a multi-scale co-association network integrating genomics, transcriptomics, and untargeted structural metabolomics at population, individual, and single-cell levels, the team identified a suite of previously unknown components involved in nicotine assembly, including a glycosyltransferase (UGT), a reductase (A622), berberine bridge enzyme-like enzymes (BBL), β-glucosidases (BGL), and a MATE transporter.
Plants, the team found, are true masters of organic chemistry. They employ an elegant and cryptic “glycosylation/deglycosylation” strategy for the final coupling reaction of nicotine’s 5- and 6-membered nitrogen-containing heterocyclic rings: This process requires two cyclic cation intermediates which are stabilized by glycosylation via a UDP-glycosyltransferase (NaUGT1), reduced and activated by an isoflavone reductase-like enzyme (NaA622), condensed through a stereoselective intermolecular Mannich-like reaction and sequentially oxidized by a berberine bridge enzyme-like (NaBBL1/2), and finally deglycosylated by a β-glucosidase (NaBGL1/2) to yield chirally pure nicotine, which is then stored in the vacuole via a MATE transporter (NaMATE1). This “nicotine synthase” process is carried out by a dynamic, five-component metabolon assembled at the vacuolar membrane. This protein complex assembly enables efficient substrate channeling, prevents the accumulation of toxic intermediates, and elegantly circumvents the “autotoxicity dilemma” inherent in plant defense.
This study completes nicotine biosynthesis, uncovering a multi-enzyme cooperative mechanism that orchestrates a stereoselective intermolecular Mannich-like reaction—a critical “scaffold-forming” step in the biosynthesis of many alkaloids. Many specialized metabolites are cytotoxic and subject to feedback inhibition, posing major bottlenecks for synthetic biology. The evolution of this integrated “biosynthesis-and-transport” metabolon allows nicotine to be efficiently produced and immediately sequestered in the vacuole, effectively neutralizing its toxicity and autoinhibition. This paradigm offers a powerful blueprint for the heterologous production of high-value natural products.
Prof. LI Dapeng is the corresponding author of the paper. The co-first authors are CHANG Lijing, XU Zhen, DENG Purong, ZHANG Ning and HE Tingrui. The research was supported by fundings from the Biological Breeding-National Science and Technology Major Project, the State Key Laboratory of Plant Trait Design, the Strategic Priority Research Program of the Chinese Academy of Sciences, the National Key Research and Development Program of China, and the National Natural Science Foundation of China.
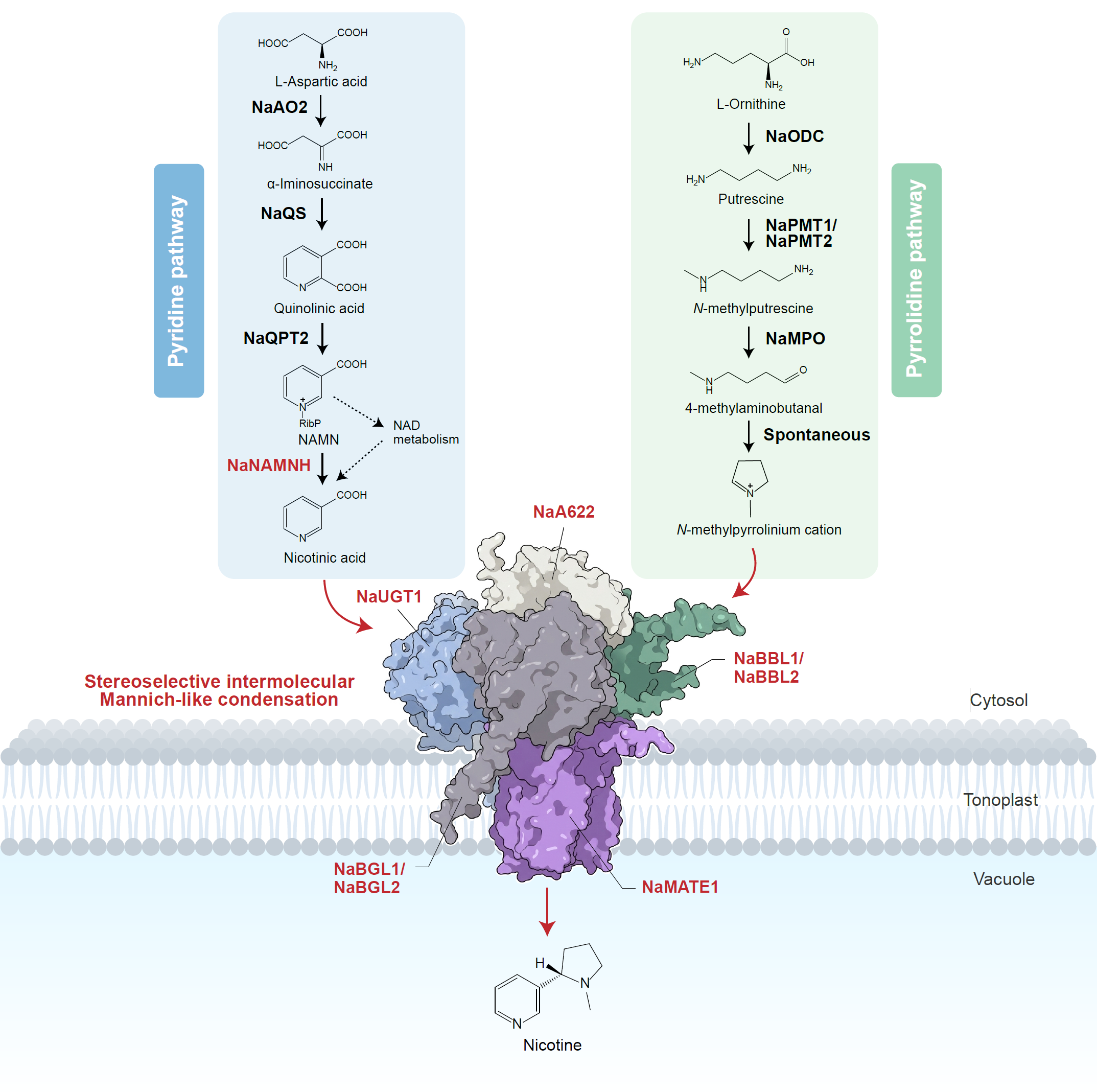
Complete biosynthesis of nicotine
Article Link: https://doi.org/10.1016/j.cell.2026.03.034
Contact: dpli@cemps.ac.cn