Iron deficiency affects two billions of people worldwide, especially children and women, and is responsible for 25,900,000 DALYs (disability-adjusted life years) and 800,000 deaths worldwide each year. The main reason for human iron deficiency is that plant seeds, as a major dietary source of iron for humans, are low in iron and high in anti-nutrition factors. Understanding the molecular basis controlling translocation of iron to the seeds are essential for resolving the global problem of human iron deficiency, as well as for plant nutrition and agricultural production.
Nicotianamine (NA) is a metal-chelating molecule required for long-distance transport of iron (Fe), as well as other mineral nutrients such as copper (Cu), in higher plants. Importantly, NA is one of the best enhancer for the bioavailability of iron in human and animals, and can help prevent hypertension and Alzheimer's disease. Identification and characterization of the genes controlling translocation of NA and NA-chelated Fe to seeds are very important to improving nutrition of human food. However, these mysterious genes and how they function have remained as long-sought questions.
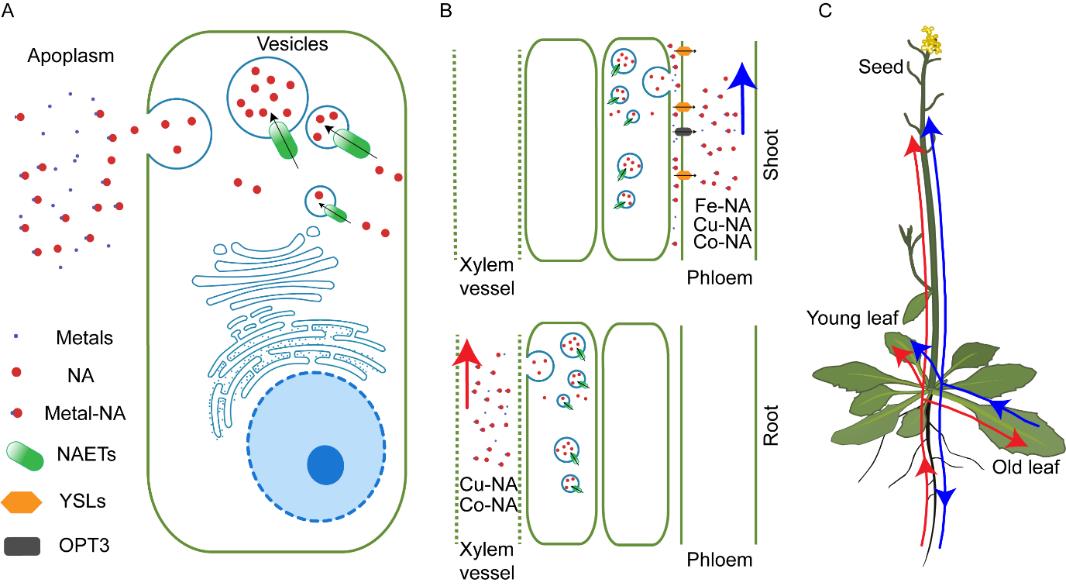
Scientists from CAS Center for Excellence in Molecular Plant Sciences (CEMPS) now reported to uncover two members of the nitrate/peptide transporter family (NPF) as NA efflux transporters, which they named NAET1 and NAET2 (NA Efflux Transporter 1 and 2), respectively. They found that NAET1 and NAET2 are in charge of unloading of NA from the cells producing them to the vascular system. Knockout of both NAET genes results in dramatic decreases of NA in the vascular system, and as well as to the sink organs including seeds and flowers. The decrease of NA in the vascular system of the mutant losing the NAET genes further leads to a 75.4% decrease of Fe and a 90.9% decrease of Cu in the seeds, demonstrating that the NAET genes play a major role in seed iron and copper contents. Further investigation revealed that the NAET1 and NAET2 proteins are localized within cells to a type of secretory vesicles, which mediates NA efflux from cells. This resembles the release of neurotransmitters from animal synaptic vesicles, which has never been found in plants.
These findings address a long-sought fundamental question in plant nutrition, and have implications for generating crops with enhanced Fe nutrition to resolve the ‘hidden hunger’ problems. The discovery of a transport mechanism in plant resembling neurotransmitter release in animal is also a significant breakthrough in plants, opening up new avenues for studying molecular and ion transport processes in plant.
This study was published on Science Advances on September 3, 2021. Prof. Dai-Yin Chao at CEMPS is the corresponding author of this paper, and PhD student Zhen-Fei Chao at CEMPS is the first author. Prof. Mingguang Lei at CEMPS and Prof. Jianbing Yan at Huanzhong Agriculture University participated in this work. This study was supported by the National Natural Science Foundation of China, the Strategic Priority Research Program of the Chinese Academy of Sciences, Sichuan Science and Technology Program and the Newton Fund.
Fig. 1. Proposed functional mechanism of NAET1 and NAET2 in metal homeostasis of A. thaliana.
Article Link: https://www.science.org/doi/10.1126/sciadv.abh2450
Contact:
Dr. Dai-Yin Chao
National Key Laboratory of Plant Molecular Genetics, CAS Center for Excellence in Molecular Plant Sciences (CEMPS), Chinese Academic of Sciences
Tel: 86-21-54924358
Email:dychao@cemps.ac.cn